
The rate at which composting occurs depends on physical as well as chemical factors. Temperature is a key parameter determining the success of composting operations. Physical characteristics of the compost ingredients, including moisture content and particle size, affect the rate at which composting occurs. Other physical considerations include the size and shape of the system, which affect the type and rate of aeration and the tendency of the compost to retain or dissipate the heat that is generated.
Temperature Curve
Compost heat is produced as a by-product of the microbial breakdown of organic material. The heat production depends on the size of the pile, its moisture content, aeration, and C/N ratio. Additionally, ambient (indoor or outdoor) temperature affects compost temperatures.
You can chart the health and progress of your compost system by taking periodic temperature measurements. A typical temperature curve for an unturned pile is shown below. How do you think that periodic turning would change this curve?
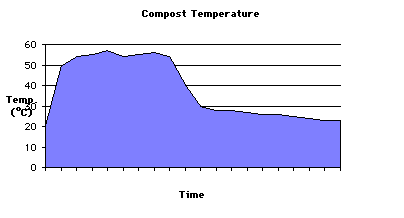
A well-designed indoor compost system, >10 gallons in volume, will heat up to 40-50°C in two to three days. Soda bottle bioreactors, because they are so small, are more likely to peak at temperatures of 30-40°C. At the other end of the range, commercial or municipal scale compost systems may take three to five days to heat up and reach temperatures of 60-70°C. Compost managers strive to keep the compost below about 65°C because hotter temperatures cause the beneficial microbes to die off. If the pile gets too hot, turning or aerating will help to dissipate the heat.
Decomposition occurs most rapidly during the thermophilic stage of composting (40-60°C), which lasts for several weeks or months depending on the size of the system and the composition of the ingredients. This stage also is important for destroying thermosensitive pathogens, fly larvae, and weed seeds. In outdoor systems, compost invertebrates survive the thermophilic stage by moving to the periphery of the pile or becoming dormant. Regulations by the U.S. Environmental Protection Agency specify that to achieve a significant reduction of pathogens during composting, the compost should be maintained at minimum operating conditions of 40°C for five days, with temperatures exceeding 55°C for at least four hours of this period. Most species of microorganisms cannot survive at temperatures above 60-65°C, so compost managers turn or aerate their systems to bring the temperature down if they begin to get this hot.
As the compost begins to cool, turning the pile usually will result in a new temperature peak because of the replenished oxygen supply and the exposure of organic matter not yet thoroughly decomposed. After the thermophilic phase, the compost temperature drops and is not restored by turning or mixing. At this point, decomposition is taken over by mesophilic microbes through a long process of "curing" or maturation. Although the compost temperature is close to ambient during the curing phase, chemical reactions continue to occur that make the remaining organic matter more stable and suitable for use with plants.
Mechanisms of Heat Loss
The temperature at any point during composting depends on how much heat is being produced by microorganisms, balanced by how much is being lost through conduction, convection, and radiation. Through conduction, energy is transferred from atom to atom by direct contact; at the edges of a compost pile, conduction causes heat loss to the surrounding air molecules.
Convection refers to transfer of heat by movement of a fluid such as air or water. When compost gets hot, warm air rises within the system, and the resulting convective currents cause a steady but slow movement of heated air upwards through the compost and out the top. In addition to this natural convection, some composting systems use "forced convection" driven by blowers or fans. This forced air, in some cases triggered by thermostats that indicate when the piles are beginning to get too hot, increases the rates of both conductive and convective heat losses. Much of the energy transfer is in the form of latent heat -- the energy required to evaporate water. You can sometimes see steamy water vapor rising from hot compost piles or windrows.
The third mechanism for heat loss, radiation, refers to electromagnetic waves like those that you feel when standing in the sunlight or near a warm fire. Similarly, the warmth generated in a compost pile radiates out into the cooler surrounding air. The smaller the bioreactor or compost pile, the greater the surface area-to-volume ratio, and therefore the larger the degree of heat loss to conduction and radiation. Insulation helps to reduce these losses in small compost bioreactors.
Moisture content affects temperature change in compost; since water has a higher specific heat than most other materials, drier compost mixtures tend to heat up and cool off more quickly than wetter mixtures, providing adequate moisture levels for microbial growth are maintained. The water acts as a kind of thermal flywheel, damping out the changes in temperature as as microbial activity ebbs and flows.
Other Physical Factors
Particle Size
Microbial activity generally occurs on the surface of the organic
particles. Therefore, decreasing particle size, through its effect
of increasing surface area, will encourage microbial activity
and increase the rate of decomposition. On the other hand, when
particles are too small and compact, air circulation through the
pile is inhibited. This decreases O2 available to microorganisms
within the pile and ultimately decreases the rate of microbial
activity.
Particle size also affects the availability of carbon and nitrogen. Large wood chips, for example, provide a good bulking agent that helps to ensure aeration through the pile, but they provide less available carbon per mass than they would in the form of wood shavings or sawdust.
Aeration
Oxygen is essential for the metabolism and respiration of aerobic
microorganisms, and for oxidizing the various organic molecules
present in the waste material. At the beginning of microbial oxidative
activity, the O2 concentration in the pore spaces is about 15-20%
(similar to the normal composition of air), and the CO2 concentration
varies form 0.5-5%. As biological activity progresses, the O2
concentration falls and CO2 concentration increases. If the average
O2 concentration in the pile falls below about 5%, regions of
anaerobic conditions develop. Providing the anaerobic activity
is kept to a minimum, the compost pile acts as a bio-filter to
trap and degrade the odorous compounds produced as a by-product
of anaerobic decomposition. However, should the anaerobic activity
increase above a certain threshold, undesireable odors may result.
Maintaining aerobic conditions can be accomplished by various methods including drilling air holes, inclusion of aeration pipes, forced air flow, and mechanical mixing or turning. Mixing and turning increase aeration by loosening up and increasing the porosity of the compost mixture.
Moisture
A moisture content of 50-60% is generally considered optimum for
composting. Microbially induced decomposition occurs most rapidly
in the thin liquid films found on the surfaces of the organic
particles. Whereas too little moisture (<30%) inhibits bacterial
activity, too much moisture (>65%) results in slow decomposition,
odor production in anaerobic pockets, and nutrient leaching. The
moisture content of compostable materials ranges widely, as shown
in the table below: 
| Material | Moisture (wet basis) |
|---|---|
| Peaches | 80% |
| Lettuce | 87% |
| Dry dog food | 10% |
| Newspaper | 5% |
Size and Shape of Compost System
A compost pile must be of sufficient size to prevent rapid dissipation
of heat and moisture, yet small enough to allow good air circulation.
A minimum of 10 gallons is required for experimental systems in
garbage cans if heat build-up is to occur within a few days. Smaller
systems can be used for classroom research or demonstration projects
but will require insulation for heat retention.
The shape of the pile helps to control its moisture content. In humid regions, outdoor compost systems may need to be sheltered from precipitation; in arid regions, piles should be constructed with a concave top to catch precipitation and any other added water.
|
Composting |
Engineering |
in Schools |
|
Cornell Waste Management Institute ©1996
Cornell University
Ithaca, NY 14853
607-255-1187
cwmi@cornell.edu